Risks related to endotoxins and their removal by using in-line IV filters
SUMMARY
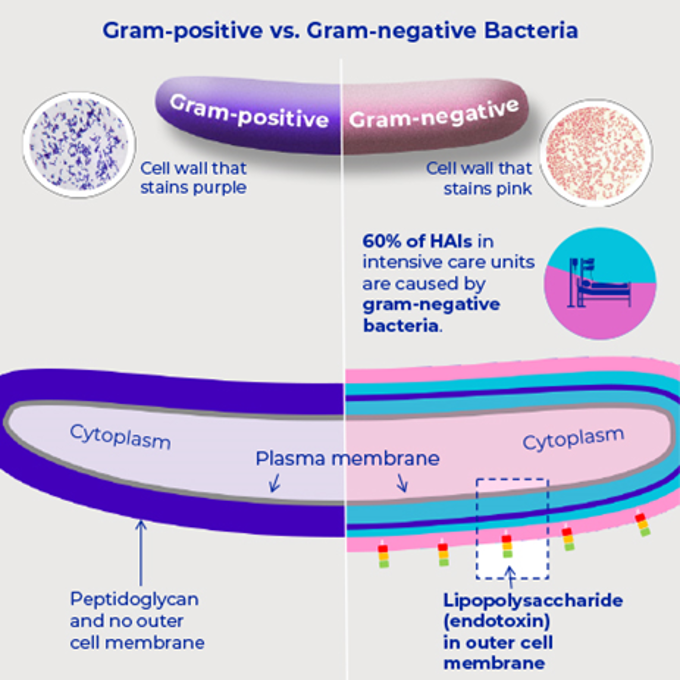
- Endotoxins, derived from gram-negative bacterial cell walls, are a key trigger of sepsis and septic shock, with even small doses causing severe systemic inflammation and multi-organ dysfunction in critically ill patients.
- Clinical investigations and validation studies show that in-line IV filters with Posidyne® membrane retain >99.9999% of endotoxins for up to 96 hours under the specifc conditions evaluated , reducing the risk of adverse reactions from contaminated IV solutions while lowering staff time and costs.
This content summarises literature and internal testing conducted under defined conditions and is not a Pall product performance or clinical outcome claim; device capabilities (including any endotoxin retention) and use conditions are defined by the applicable Instructions for Use.
Why are endotoxins dangerous in IV therapy, and how can in-line filters help prevent harm?
The investigation of the function of endotoxin in the pathophysiology of sepsis has been studied since the 1800s, when endotoxin was first revealed as a gram-negative cell wall toxin responsible for lethal shock.1 Today, systemic gram-negative sepsis with endotoxin as the prime initiator remains one of the most severe conditions complicating the course of hospitalized subjects, mainly in critically ill patients in the intensive care unit (ICU).2,3
Chemical structure analysis has shown an endotoxin unit is consisting of a lipid A component and an O-Antigen component. The O-Antigen is made of polysaccharides and is associated with immunogenicity, while the lipid A portion of the endotoxin is the toxic component.4 The immune system's reaction to a lipid A component may lead to serious generalized inflammation, manifesting clinically as septic shock with multiple organ dysfunction, especially myocardial depression and renal impairment.5 In IV therapy gram-negative bacteria and associated endotoxin may get into the patient’s blood stream from an external source, such as contaminated equipment or fluids.6-13 Hospital cases of endotoxin in intravenous solutions leading to adverse reactions or even death have been investigated in the past.14-19
This is where our in-line IV filters with Posidyne membrane come into play - retaining endotoxin and preventing it entering the patient’s bloodstream during IV therapy. Our validation testing over 96 hours confirmed that:
- Our ELD96 in-line IV filters (aged and unaged) retained > 99.9999 % of the endotoxin challenge produced from a 1 x 108 E. coli challenge in 0.9% saline with an effluent concentration of < 0.1 EU/mL* from an average challenge level of > 1 x 103 EU/cm2 (*limit of detection of the assay = 0.1 EU/mL).20
- Our NEO96 in-line IV filters (aged and unaged) retained > 99.9999 % of the endotoxin challenge produced from a 1 x 108 E. coli challenge in 0.9% saline with an effluent concentration of < 0.1 EU/mL* from an average challenge level of > 1 x 104 EU/cm2 (*limit of detection of the assay = 0.1 EU/mL).21
If a patient is already sick, with septic shock or other critical illness, will a few endotoxin units really make a difference? Yes, studies have described the clinical effects of even small doses of endotoxin on humans.22,23
What to consider when evaluating in‑line filtration
- Retention of endotoxin up to 96 hours under clinically relevant conditions 24-27
- Reducing staff time & costs due to less IV filter-set changes 28-30
- Improvement of clinical outcomes 32-36
- We are the leading pioneer and have manufactured positively charged, membrane-based in-line IV filter products with a claim for the removal of bacteria and associated endotoxins for the health care community for over four decades
Explore our IV endotoxins removal solutions
References
1.Romaschin A.D., Klein D.J., Marshall JC. (2012). Bench-to-bedside review: Clinical experience with the endotoxin activity assay. Crit Care; 16 (6): 248
2.Sakr Y, Jaschinski U, Wittebole X, et al. (2018). Sepsis in Intensive Care Unit Patients: Worldwide Data From the Intensive Care over Nations Audit. Open Forum Infect Dis;5 (12): 1-9
3.Ianaro A, Tersigni M, D'Acquisto F. (2009) New insight in LPS antagonist. Mini Rev Med Chem; 9 (3):306-17.
4.Sampath VP. (2018). Bacterial endotoxin-lipopolysaccharide; structure, function and its role in immunity in vertebrates and invertebrates. Agriculture and Natural Resources; 52 (2): 115-120
5.Virzì G.M. et al. (2017). Endotoxin Effects on Cardiac and Renal Functions and Cardiorenal Syndromes. Blood Purif; 44: 314-326
6.Twum-Danso K, Dawodu AH, Saleh M.A.F., Makiling L.S. (1989). An out-break of K. pneumoniae bacteremia in five children on intravenous therapy. J. Hosp. Infest; 14: 271–274.
7.Ng P.C. et al. (1989). An outbreak of Acinetobacter septicaemia in a neonatal intensive care unit. Journal of Hospital Infection; 14: 363-368
8.Lacey S. & Want S.V. (1991). Pseudomonas pickettii infections in a paediatric oncology unit. Journal of Hospital Infection; 17 (1): 45-51
9.Ezzedine H. et al. (1994). An outbreak of Ochrobactrumanthropi bacteremia in five organ transplant patients. J Hosp Infect; 27: 35-42
10.J.A.Frean, Arntzen L., Rosekilly I., Isaäcson M. (1994). Investigation of contaminated parenteral nutrition fluids associated with an outbreak of Serratia odorifera septicaemia. Journal of Hospital Infection; 27 (4): 263-273
11.Bernards A.T. et al. (1997). Outbreak of septicaemia in neonates caused by Acinetobacter junii investigated by amplified ribosomal DNA restriction analysis (ARDRA) and four typing methods. Journal of Hospital Infection; 35 (2): 129-140
12.Garland S.M. et al. (1996). Pseudomonas aeruginosa outbreak associated with a contaminated blood-gas analyser in a neonatal intensive care unit. Journal of Hospital Infection; 33: 145-151
13.Holmes C.J. et al. (1980). Potential Hazards Associated with Microbial Contamination of In-Line Filters During Intravenous Therapy. Journal Of Clinical Microbiology; 12 (6): 725-7:31
14.Garrett D.O. et al. (2002). An Outbreak of Neonatal Deaths in Brazil Associated with Contaminated Intravenous Fluids. The Journal of Infectious Diseases; 186 (1): 81–86
15.Daufenbach, L. (2006). Pyrogenic Reactions and Hemorrhage Associated With Intrinsic Exposure to Endotoxin-Contaminated Intravenous Solutions. Infection Control & Hospital Epidemiology; 27 (7): 735-741
16.Schroeder J. et al. (2015). Practically Saline. Journal of Investigative Medicine High Impact Case Reports; 1-4
17.CDC (1998). Endotoxin-Like Reactions Associated with Intravenous Gentamicin -- California, 1998. Retrieved from: https://www.cdc.gov/mmwr/preview/mmwrhtml/00055322.htm
18.Johnstone T. et al. (2018). Seven cases of probable endotoxin poisoning related to contaminated glutathione infusions. Epidemiol Infect. 2018;146(7):931-934. doi:10.1017/S0950268818000420
19.Patel AS, et al. (2006) Outbreak of systemic inflammatory response syndrome linked to a compounding pharmacy – Virginia, 2005 In. 55th Annual Epidemic Intelligence Service Conference. Atlanta, Georgia, USA: U.S. Department of Health and Human Services.
20.Ragunath S. & Spiers S. (2021). Evaluation of endotoxin retention efficiency of Pall ELD96 IV filters with 0.2 µm Posidyne® membrane over a 96-hour period; Pall Technical Report
21.Ragunath S. & Spiers S. (2021). Evaluation of endotoxin retention efficiency of Pall NEO96 IV filters with 0.2 µm Posidyne® membrane over a 96-hour period; Pall Technical Report
22.Suffredini A.F., Hochstein H.D., McMahon F.G. (1999). Dose-related inflammatory effects of intravenous endotoxin in humans: evaluation of a new clinical lot of Escherichia coli O:113 endotoxin. J Infect Dis; 179 (5): 1278-82
23.Bahador M., Cross A.S. (2007). From therapy to experimental model: a hundred years of endotoxin administration to human subjects. Journal of Endotoxin Research; 13 (5): 251-279
24.Baumgartner, T. G. et al. (1986). Bacterial endotoxin retention by inline intravenous filters. Am. J. Hosp. Pharm; 43:681-684
25.Horibe, K. et al. (1990). Evaluation of the endotoxin retention capabilities of inline intravenous filters. JPEN J. Parenter. Enteral. Nutr; 14: 56-59
26.Richards, C. & Grassby P. F. (1994). A comparison of the endotoxin-retentive abilities of two ‘96-h’ in-line intravenous filters. J. Clin. Pharm. Ther; 19 (3): 199-202
27.Spielberg, R., and J. Martin. 1985. Evaluation of the endotoxin/bacterial retention of I.V. filters during simulated extended infusions, p. 1001. In Technical note IV. Pall Biomedical Ltd., Portsmouth, United Kingdom.
28.Villa G. et al. (2020). In-line filtration reduced phlebitis associated with peripheral venous cannulation: Focus on cost-effectiveness and patients' perspectives. J Vasc Access; 21(2): 154-160
29.Van den Hoogen A. et al. (2006). In-line filters in central venous catheters in a neonatal intensive care unit. J Perinat Med; 34(1): 71-4
30.Van Lingen et al. (2004). The use of in-line intravenous filters in sick newborn infants. Acta Paediatr; 93(5): 658-62
31.Unger-Hunt L. (2019). Reducing Risks and Generating Economic Benefits. Health Management; 19 (4): 286-287
32.Jack T. et al. (2012). In-line filtration reduces severe complications and length of stay on pediatric intensive care unit: a prospective, randomized, controlled trial. Intensive Care Med; 38(6): 1008-16
33.Boehne M. et al. (2013). In-line filtration minimizes organ dysfunction: New aspects from a prospective, randomized, controlled trial. BMC Pediatrics; 13 (21): 1-8
34.Sasse M. et al. (2015). In-line Filtration Decreases Systemic Inflammatory Response Syndrome, Renal and Hematologic Dysfunction in Pediatric Cardiac Intensive Care Patients. Pediatr Cardiol; 36: 1270-1278
35.Villa G. et al. (2018). In-Line Filtration Reduces Postoperative Venous Peripheral Phlebitis Associated With Cannulation: A Randomized Clinical Trial. Anesth Analg; 127(6): 1367-1374
36.Virlouvet A.L. et al. (2020). In-line filtration in very preterm neonates: a randomized controlled trial. Scientific Reports; 10 (5003): 1-8
Author bio
Dr. Luibl is a Sr. Marketing Manager Medical Content with knowledge in medical device and clinical science.