Even small doses of endotoxin can lead to clinical effects in humans
SUMMARY
Endotoxin exposure can cause fever, tachycardia, hypotension, vascular leakage, and coagulation abnormalities, progressing to multi-organ failure in severe cases. Experimental studies show that as little as 2–4 ng/kg (20-40 EU/kg) induces fever, increased heart rate, and inflammatory responses in healthy volunteers.
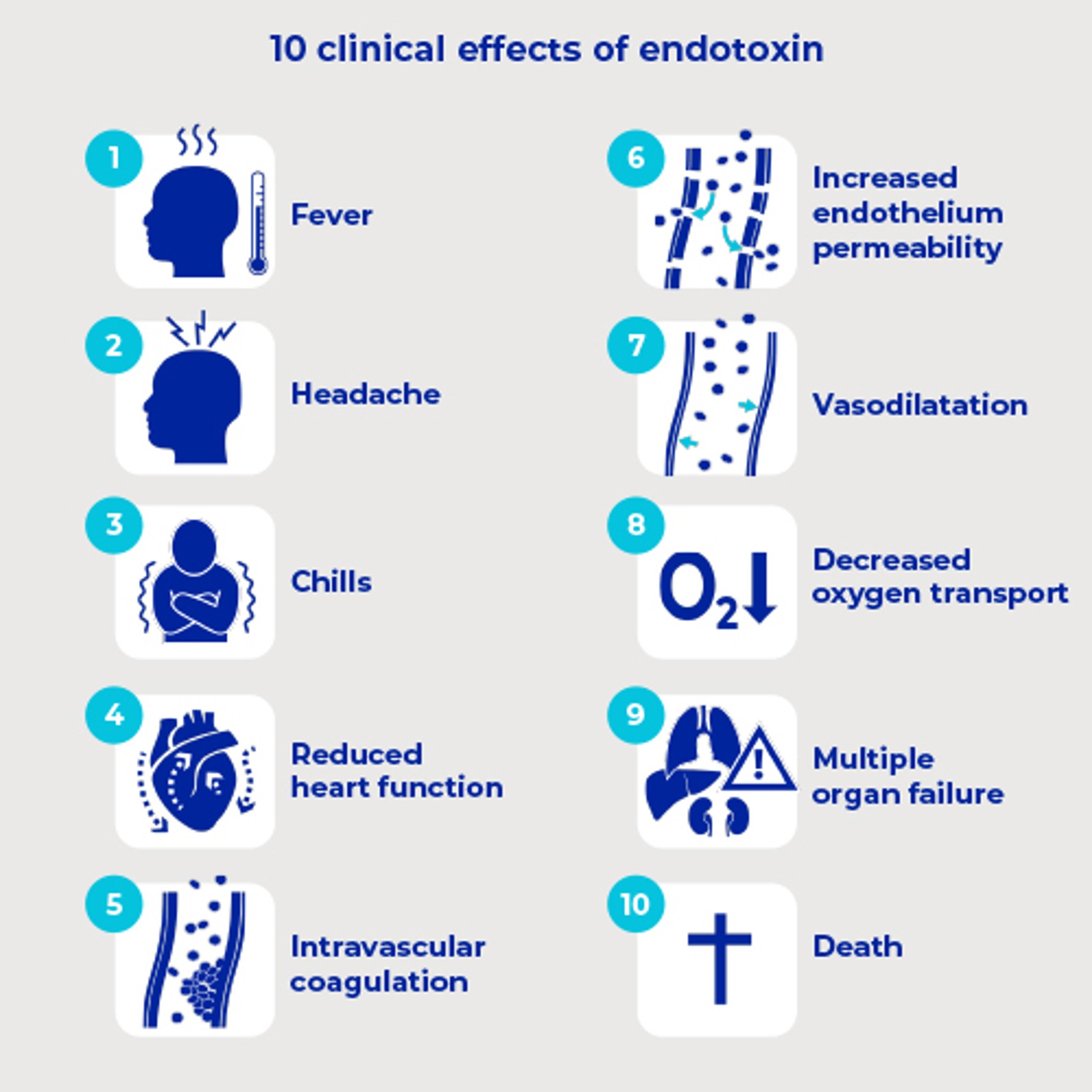
What are the clinical effects of even small doses of endotoxin in humans?
The clinical consequences of endotoxin include1,2 :
- fever, headache, chills
- reduced heart function (cardiac contractility), causing tachycardia to try to keep the circulation going
- intravascular coagulation, leading to vascular occlusion (blockage of circulation)
widening of the blood vessels (vasodilatation), causing a fall in blood pressure (hypotension) - damage to the lining of the blood vessels (increased endothelium permeability), causing leakage of fluid from the blood vessel into the surrounding tissue (oedema)decreased oxygen transport to the tissues (decreased perfusion)in the worst cases, the heart is unable to pump blood around the system, the blood pressure falls, large areas of circulation are not reached, especially in the organs that require a high throughput of blood, and the tissues don't get enough oxygen.
Due to the above, there may be progressive loss of function in the kidneys, lungs, heart, and brain, leading to multiple organ failure, which can be fatal.
Studies have described the effects of even small doses of endotoxin on humans. Experiments on healthy volunteers found adverse effects using 2-4 nanograms of endotoxin per kilogram body weight. This equates to 20-40 endotoxin units (EU) per kg. At this level, the subjects experienced fever, increased heart rate, headache, chills, aching limbs, and were found to have significantly decreased cardiac function, increased blood pressure, raised white cell count and inflammatory factors, increased gut permeability and activation of the coagulation system.3-10
Explore our IV endotoxins removal solutions
References
1.Glauser M.P., Zanetti G., Baumgartner J.D., Cohen J. (1991). Septic shock: pathogenesis. Lancet; 338 (8769): 732-6
2.Balk R.A. (1994). Septic shock: pathophysiology. Current Opinion in Anesthesiology; 7(2): 136-140
3.Fullerton J.N. et al. (2016). Intravenous Endotoxin Challenge in Healthy Humans: An Experimental Platform to Investigate and Modulate Systemic Inflammation. J Vis Exp; (111): 53913
4.Calvano SE, Coyle SM. (2012): Experimental human endotoxemia: a model of the systemic inflammatory response syndrome? Surgical infections;13(5): 293–299
5.Bahador M., Cross A.S. (2007). From therapy to experimental model: a hundred years of endotoxin administration to human subjects. Journal of Endotoxin Research; 13 (5): 251-279
6.Suffredini A.F., Hochstein H.D., McMahon F.G. (1999). Dose-related inflammatory effects of intravenous endotoxin in humans: evaluation of a new clinical lot of Escherichia coli O:113 endotoxin. J Infect Dis; 179 (5): 1278-82
7.Suffredini A.F. et al. (1989). The Cardiovascular Response of Normal Humans to the Administration of Endotoxin. N Engl J Med; 321: 280-287
8.Casale TB et al. (1990). The effects of intravenous andutoxin on various host-effector molecules. J Allergy Clinical Immunology; 85: 45-51
9.Suffredini A.F., Harpel P.C., Parrillo J.E. (1989). Promotion and Subsequent Inhibition of Plasminogen Activation after Administration of Intravenous Endotoxin to Normal Subjects. N Engl J Med; 320: 1165-1172
10.Gralnick H.R. et al. (1989). Von Willebrand factor release induced by endotoxin. J Laboratory Clinical Medicine; 113 (1): 118-122
Author bio
Dr. Luibl is a Sr. Marketing Manager Medical Content with knowledge in medical device and clinical science.