Principal mechanisms of IV filtration
Inside an in-line IV filter: learn more about IV filtration mechanisms.
The problem
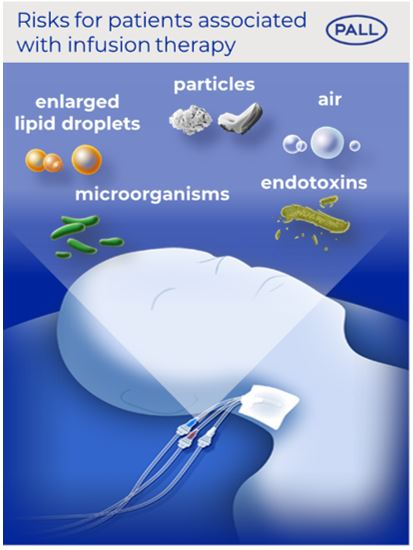
Intravenous drug infusions can be a life-saving therapy. It has been shown that these solutions may contain inadvertent particles, ambient air, microorganisms, their associated endotoxins, and enlarged lipid droplets which can potentially put the patient’s health at risk 1-4.
Our solution: Experimental and clinical studies have proven that our in-line IV filtration devices can retain particles, air bubbles, microorganisms, their associated endotoxins, and enlarged lipid droplets 5-7.
Read here about the mechanisms of in-line IV filtration and how in-line IV filter retain particles, air bubbles, microorganisms, their associated endotoxins, and enlarged lipid droplets.
Mechanisms of particle filtration
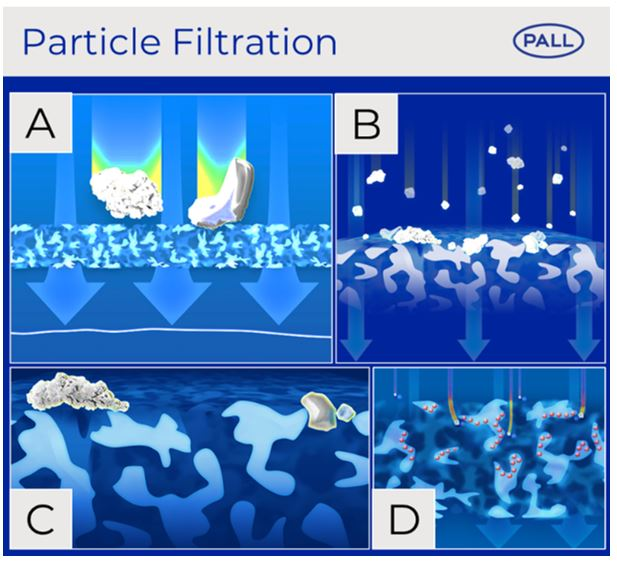
Particulates above 10 µm in solutions for intravenous therapy are regulated by European and US Pharmacopeias 4. However, smaller particles and particles inadvertently produced during preparation and administration of the solutions to the patient are not/cannot be regulated. Intensive care patients who receive a multitude of IV infusions can therefore be infused with up to one million particles per day in the absence of in-line IV filters 8-11. Infused particles could lead to a disturbance in the microcirculation and compromised microvascular flow in vital organs resulting in organ dysfunction of ICU patients9-12.
Ourin-line IV filters retain particles by conventional mechanisms of direct interception which include size exclusion (A), cake filtration (B) and bridge filtration (C). Additionally, particulates may be retained by inertial impaction within the membrane or diffusion Interception where there are charge effects (D).
Mechanisms of air filtration
Estimates of the frequency of vascular air embolism (VAE) related to central venous catheters (CVCs) vary from study to study and are reported to range from 1 in 47 to 1 in 3000 catheterization events or from 0.1% to 2% per patient (13-15). While the frequency of this complication may be low, mortality rates attributed to venous air embolisms associated with CVCs range from 23% to 50% 16-20.
Hydrophobic vent membranes on our in-line IV filters allow effective air elimination that may be entrained in infusions (i.e., due to degassing, disconnection, etc.) and protect against air emboli in patients.
Mechanisms of microorganisms filtration

Lipid-containing parenteral nutrition (PN) solutions are often considered as an ideal microbial growth medium, and slow administration at room temperature offers the opportunity for microbes to multiply and cause adverse effects 21-23.
Our 1.2 µm IV filters have the possibility to retain inadvertent microbes with a size above 1.2 µm, such as Candida species. Our 0.2 µm IV filters can retain smaller inadvertent microbes, which could include Staphylococcus epidermidis and Escherichia coli.
Mechanisms of endotoxin filtration
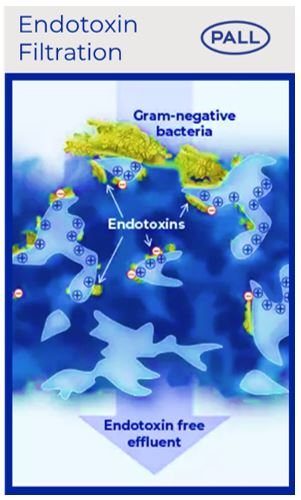
Previous studies have shown that gram-negative bacteria can multiply and will shed endotoxin in intravenous solutions 23-24. Inadvertent infusion of nonsterile fluids contaminated with endotoxins may be rare, but hospital cases of endotoxin in intravenous solutions leading to adverse reactions or even death have been investigated 25-29.
Our Posidyne™ positively charged membrane (+) retains endotoxin due to the binding to the negative charge of endotoxins (-).
Filtration of enlarged lipid droplets
Maintaining stability of lipid-containing parenteral nutrition solutions is critical, since destabilization can cause the lipid globules to coalesce, thus causing the particle size to exceed 5 µm. If particle size exceeds 5 µm patients can be at risk of pulmonary capillary occlusion and fat emboli 30.
Our 1.2 µm IV filters have the possibility to retain lipid droplets with a size above 1.2 µm.
With a strong focus on patient safety and satisfaction, our expertise on in-line IV filtration devices optimizes infusion therapy worldwide.
Explore our IV filtration solutions
FAQs
What risks do in-line IV filters address?
They retain particles, air bubbles, microorganisms, endotoxins, and enlarged lipid droplets that can harm patients during infusion therapy. 1-4
How do filters remove particles?
Through size exclusion, cake filtration, bridge filtration, inertial impaction, and charge-based interception. 5-12
How do IV filters prevent air embolism?
Hydrophobic vent membranes eliminate entrained air, protecting patients from vascular air embolism. 13-20
Can IV filters retain microorganisms and endotoxins?
Yes. 1.2 µm filters capture larger microbes like Candida, while 0.2 µm filters retain smaller microbes. Posidyne™ membranes bind endotoxins via charge interaction. 21-29
Why is lipid droplet filtration important?
Droplets >5 µm can block pulmonary capillaries, causing fat emboli. 1.2 µm filters retain enlarged lipid droplets to prevent complications. 30
References
- Myers G.J. (2017). Air in intravenous lines: a need to review old opinions. Perfusion; 32 (6): 432-435
- F. Merry A.F., Gargiulo D.A., Fry L.E. (2017). What are we injecting with our drugs? Anaesth Intensive Care; 45 (5): 539-542
- Holmes C.J., Kundsin R.B., Ausman R.K., Walter C.W. (1980). Potential hazards associated with microbial contamination of in-line filters during intravenous therapy. J Clin Microbiol;12 (6): 725-731
- Perez M., Maiguy-Foinard A., Barthélémy C., Décaudin B., Odou P. (2016). Particulate Matter in Injectable Drugs: Evaluation of Risks to Patients. Pharm. Technol. Hosp. Pharm.; 1(2): 91-103
- Perez M. et al. (2018). Effectiveness of in-Line Filters to Completely Remove Particulate Contamination During a Pediatric Multidrug Infusion Protocol. Sci Rep; 8 (7714): 1 - 8
- Van Lingen R.A., Baerts W., Marquering A.C., Ruijs G.J. (2004). The use of in-line intravenous filters in sick newborn infants. Acta Paediatr; 93 (5): 658-62
- Richards C., Grassby P.F. (1994). A comparison of the endotoxin-retentive abilities of two '96-h' in-line intravenous filters. J Clin Pharm Ther;19 (3): 199-202
- Perez M. et al. (2015). In vitro analysis of overall particulate contamination exposure during multidrug IV therapy: impact of infusion sets. Pediatr Blood Cancer; 62(6): 1042-7
- Benlabed M. et al. (2019). Clinical implications of intravenous drug incompatibilities in critically ill patients. Anaesth Crit Care Pain Med;38(2): 173-180.
- Lehr HA., Brunner J., Rangoonwala R. and Kirkpatrick C.J. (2002). Particulate Matter Contamination of Intravenous Antibiotics Aggravates Loss of Functional Capillary Density in Postischemic Striated Muscle. Am J Respir Crit Care Med; 165: 514-520
- Kirkpatrick CJ. et al. (2013). Non-Equivalence of Antibiotic Generic Drugs and Risk for Intensive Care Patients. Pharmaceut Reg Affairs; 2(1): 1-7
- Schaefer S.C., Bison P.A., Rangoonwala R., Kirkpatrick J.C. and Lehr H.A. (2008). 0.2 µm in-line filters prevent capillary obstruction by particulate contaminants of generic antibiotic preparations in postischemic muscle. Chemother J; 17: 172-8
- Cook L.S. (2013). Infusion-Related Air Embolism. Journal of Infusion Nursing; 36 (1): 26-36
- Boersma R.S., Jie K.S., Verbon A., van Pampus E.C., Schouten H.C. (2008). Thrombotic and infectious complications of central venous catheters in patients with hematological malignancies. Ann Oncol; 19: 433-442
- Gordy S., Rowell S. (2013). Vascular air embolism. Int J Crit Illn Inj Sci;3 (1): 73-76
- Scott W.L. (1988). Complications Associated with Central Venous Catheters: A Survey. Chest; 94 (6): 1221-1224
- Vesely T.M. (2001). Air Embolism during Insertion of Central Venous Catheters. Journal of Vascular and Interventional Radiology; 12 (11): 1291-129
- Feil M. (2015). Preventing central line air embolism. Am J Nurs; 115 (6): 64-9.
- Heckmann J.G. et al. (2000). Neurologic manifestations of cerebral air embolism as a complication of central venous catheterization. Crit Care Med; 28(5): 1621-5
- Kashuk J.L., Penn I. (1984). Air embolism after central venous catheterization. Surg Gynecol Obstet; 159: 249-52
- Austin P.D. et al. (2016). Systematic review and meta-analyses of the effect of lipid emulsion on microbial growth in parenteral nutrition. Journal of Hospital Infection; 94(4): 307-319
- Holmes C.J. et al. (1980). Potential Hazards Associated with Microbial Contamination of In-Line Filters During Intravenous Therapy. Journal of Clinical Microbiology; 12 (6): 725-731
- Trautmann M. et al. (1997). Bacterial colonization and endotoxin contamination of intravenous infusion fluids. J Hosp Infect; 37(3): 225-36
- Jorgensen J.H. and Smith R.F. (1973). Rapid detection of contaminated intravenous fluids using the Limulus in vitro endotoxin assay. Appl Microbiol; 26(4): 521-524
- Garrett D.O. et al. (2002). An Outbreak of Neonatal Deaths in Brazil Associated with Contaminated Intravenous Fluids. The Journal of Infectious Diseases; 186 (1): 81–86
- Daufenbach, L. (2006). Pyrogenic Reactions and Hemorrhage Associated With Intrinsic Exposure to Endotoxin-Contaminated Intravenous Solutions. Infection Control & Hospital Epidemiology; 27 (7): 735-741
- Schroeder J. et al. (2015). Practically Saline. Journal of Investigative Medicine High Impact Case Reports; 1-4
- Endotoxin-Like Reactions Associated with Intravenous Gentamicin --California, 1998. Retrieved from: https://www.cdc.gov/mmwr/preview/mmwrhtml/00055322.htm
- Johnstone T. et al. (2018). Seven cases of probable endotoxin poisoning related to contaminated glutathione infusions. Epidemiol Infect. 2018;146(7):931-934. doi:10.1017/S0950268818000420
Driscoll DF. Lipid injectable emulsions: Pharmacopeial and safety issues. Pharm Res. 2006 Sep;23(9):1959-69.
Author bio
Dr. Volker Luibl, MBA
Dr. Luibl is a Sr. Marketing Manager Medical Content with knowledge in medical device and clinical science.