Evaluation of the multiplication of endotoxins in IV solutions
SUMMARY
- Gram-negative bacteria can multiply in IV solutions and release endotoxins, with documented hospital outbreaks linking contaminated fluids to adverse reactions and deaths.
- Testing made with E. coli have shown that the level of endotoxins start to increase after 24 hours and rise rapidly to become stable over 96 hours under the specific conditions evaluated.
Endotoxin levels in IV solutions can rise rapidly, creating risks for patients.
Previous studies have shown that gram-negative bacteria can multiply and will shed endotoxin in intravenous solutions.1-3 Accidental infusion of nonsterile fluids contaminated with endotoxins may be rare, but hospital cases of endotoxin in intravenous solutions leading to adverse reactions or even death have been investigated.4-9
Our test: The accumulation of endotoxin in infusion solutions
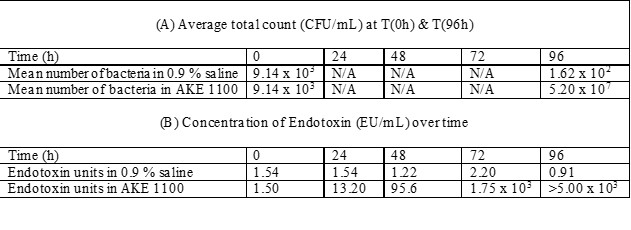
We have previously monitored the concentration of E.coli and corresponding endotoxin levels over a 96-hour period when a 1 L container of 0.9 % Saline or 1 L of AKE 1100 with Xylit (Fresenius Kabi) was contaminated with 1 x 108 CFU of E.coli.
The table below shows the concentration of E.coli (CFU/mL) detected in the solutions at time 0 and following 96 hours storage at ambient temperature (internal data).
While the endotoxin units / mL remains relatively constant over 96 hours in saline solution, endotoxin units / mL increased in the parenteral nutrition solution approximately
- by a factor of 10 after 24 hours,
- by a factor of 100 after 48 hours,
- by a factor of 1,000 after 72 hours,
- by a factor > 1,000 after 96 hours (above the limit of the assay).
Explore our IV endotoxins removal solutions
References
1.Holmes C.J. et al. (1980). Potential Hazards Associated with Microbial Contamination of In-Line Filters During Intravenous Therapy. Journal of Clinical Microbiology; 12 (6): 725-731
2.Trautmann M. et al. (1997). Bacterial colonization and endotoxin contamination of intravenous infusion fluids. J Hosp Infect; 37(3): 225-36
3.Jorgensen J.H. and Smith R.F. (1973). Rapid detection of contaminated intravenous fluids using the Limulus in vitro endotoxin assay. Appl Microbiol; 26 (4): 521-524
4.Garrett D.O. et al.(2002). An Outbreak of Neonatal Deaths in Brazil Associated with Contaminated Intravenous Fluids. The Journal of Infectious Diseases; 186 (1): 81–86
5.Daufenbach, L. (2006). Pyrogenic Reactions and Hemorrhage Associated With Intrinsic Exposure to Endotoxin-Contaminated Intravenous Solutions. Infection Control & Hospital Epidemiology; 27 (7): 735-741
6.Schroeder J. et al. (2015). Practically Saline. Journal of Investigative Medicine High Impact Case Reports; 1-4
7.CDC (1998). Endotoxin-Like Reactions Associated with Intravenous Gentamicin -- California, 1998. Retrieved from: https://www.cdc.gov/mmwr/preview/mmwrhtml/00055322.htm
8.Johnstone T. et al. (2018). Seven cases of probable endotoxin poisoning related to contaminated glutathione infusions. Epidemiol Infect. 2018;146(7):931-934. doi:10.1017/S0950268818000420
9.Patel AS, et al. (2006) Outbreak of systemic inflammatory response syndrome linked to a compounding pharmacy – Virginia, 2005 In. 55th Annual Epidemic Intelligence Service Conference. Atlanta, Georgia, USA: U.S. Department of Health and Human Services.
Author bio
Dr. Luibl is a Sr. Marketing Manager Medical Content with knowledge in medical device and clinical science.